Fragment Analysis Settings
After PCR amplification, the Non-T and Non-B Genomic Detection kits amplification products are analyzed by capillary electrophoresis on a sequencer (genetic analyser).
Instrument Settings
On Applied Biosystems instruments (such as the 3500, 3730, SeqStudio Genetic Analysers), the settings used with the chimerism/identity testing kit are usually compatible with Accumol's kits. The kits amplification products are labeled with 6-FAM. Typically, choosing an internal size standard labeled with LIZ (with filter set G5) or ROX (with filter set D) will give good results.
Sample Preparation
Mix 0.5µL of amplification product, 0.5 µL of internal size standard, 9 µL of formamide. Denature 3 minutes at 95ºC, then immediately transfer on ice. Keep on ice until ready to load the sequencer.
GeneMapper panel and bin files
These files can be imported into GenMapper 4.1 or newer versions, as well as into the Microsatellite Analysis (MSA) online application on the ThermoFisher cloud. Since the calculated size of the amplicons can vary slightly between instruments and experimental conditions, the bin and panel sizes might need to be adjusted to ensure proper peak identification.
Non-T Genomic Detection Kit: Panel file Bin file
Non-B Genomic Detection Kit: Panel file Bin file
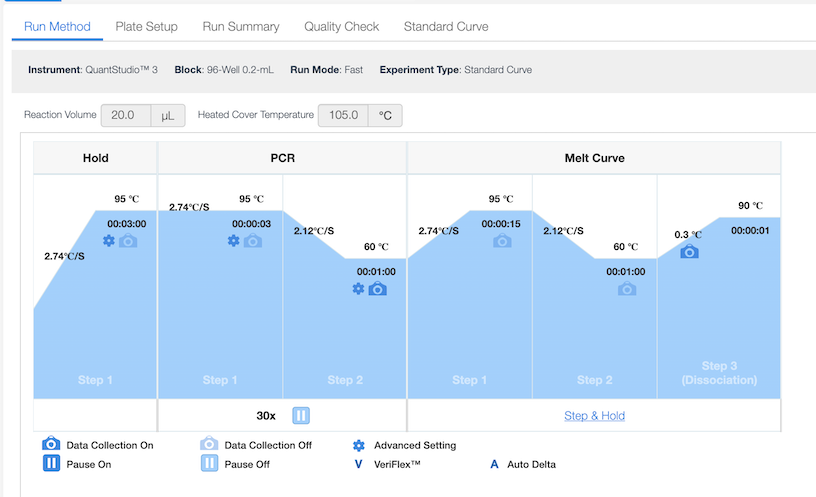
qPCR Settings
The typical settings for Applied Biosystems QuantStudio instruments are shown below. For other instruments, please contact technical support at support@accumol.com.
6-FAM, GeneMapper, SeqStudio, QuantStudio, are registered trademarks of Applied Biosystems Inc
Quick Links
- Non-T Genomic Detection Kit
- Non-B Genomic Detection Kit
- Non-Myeloid Genomic Detection Kit
- MTBN Purity Assessment Kit
- Applications
- Order Now
News
New Publication
March 2021
HLA. 2021;97:183-187
A one‐step assay for sorted CD3+ cell purity and chimerism after hematopoietic stem cell transplantation.
Desoutter et al.
New Publication
December 2019
Biomed J Sci & Tech Res 23(5)-2019
Assessment of the Purity of Isolated CD3+ T Cell Populations by A New Molecular Biology Technology: Impact on the Quantification of Chimerism Monitoring After Hematopoietic Stem Cell Transplantation.
Pedini et al.
qPCR kits released!
April 25, 2017
The Non-T and Non-B Genomic Detection kits are now available for quantitative PCR (real-time PCR) instruments. In addition, we are pleased to introduce the brand new qPCR Non-Myeloid Genomic Detection kit, designed to assess the purity of Myeloid cell preparations directly at the DNA level.
New Publication
January 2015
British Journal of Haematology, 2015, 168, 26–37
